Chemical Periodic Table – How It Is Useful for Chemistry Students (Class 9th to 12th)
Chemical Periodic Table – How It Is Useful for Chemistry Students (Class 9th to 12th)
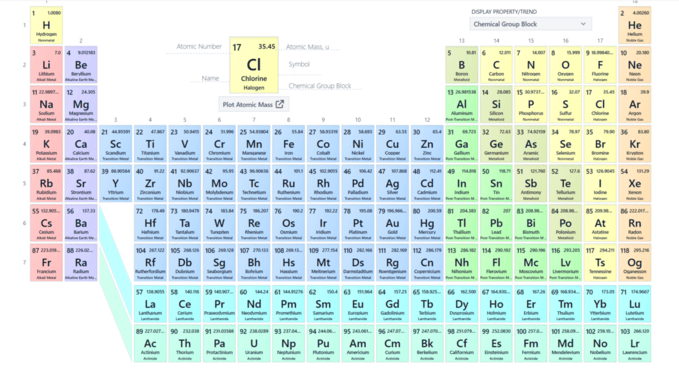
The periodic table is one of the most important tools in chemistry. Whether you are studying in class 9, 10, 11 or 12, understanding the periodic table makes learning chemistry easier, logical and interesting. It is not just a chart of elements—it is like a map of chemistry that helps you know everything about elements, from their structure to their reactions.
⭐ What Is the Periodic Table?
The periodic table is a scientifically arranged chart of 118 elements based on:
- Atomic number (number of protons)
- Electronic configuration
- Chemical properties
- Physical properties
Elements are arranged in Periods (rows) and Groups (columns) so that students can compare properties easily.
⭐ Why Is the Periodic Table Important for Students?
- ✔ Helps in Learning Atomic Number and Symbol
Instead of memorizing randomly, the table helps you learn atomic numbers in order:
Element | Symbol | Atomic Number |
Hydrogen | H | 1 |
Helium | He | 2 |
Lithium | Li | 3 |
This is useful for:
✔ Formula writing
✔ Chemical equation balancing
✔ Competitive exams
- ✔ Predict Chemical Properties
Students can easily find:
- Metals and non-metals
- Highly reactive elements
- Noble gases
- Halogens
- Transition metals
Example:
👉 Sodium (Na) and Potassium (K) react strongly with water
👉 Noble gases like Helium (He) and Neon (Ne) are unreactive
You don’t need to memorize—just look at their position.
- ✔ Understand Trends Easily
The periodic table shows important trends:
🔹 Atomic size
🔹 Electronegativity
🔹 Ionization energy
🔹 Metallic character
For example:
➡ Atomic size increases from top to bottom
➡ Atomic size decreases from left to right
This helps in explaining:
✔ Why Cesium is more reactive than Sodium
✔ Why Fluorine is more reactive than Chlorine
- ✔ Helps in Electronic Configuration
Using the table, students can write electron distribution easily.
Example:
Carbon (6)
→ 2 + 4 = K(2), L(4)
Magnesium (12)
→ 2 + 8 + 2 = K(2), L(8), M(2)
This is useful for:
✔ Formation of chemical bonds
✔ Ionic and covalent compounds
✔ Understanding valency
- ✔ Recognize Groups and Periods
The periodic table contains:
- 18 Groups
- 7 Periods
Group 1 → Alkali metals
Group 2 → Alkaline earth metals
Group 17 → Halogens
Group 18 → Noble gases
Students learning bonding and reactions find this very helpful.
⭐ How Useful Is the Periodic Table for Class 9-12 Students?
✔ Class 9 & 10 Students
- Basic element classification
- Metals, non-metals & metalloids
- Physical & chemical properties
- Modern periodic table concept
✔ Class 11 & 12 Students
- Atomic structure
- Periodicity trends
- s-block, p-block, d-block & f-block chemistry
- Bonding and reactivity
- Numerical chemistry
⭐ Memory Tricks for Learning Groups Easily
👉 H Li Na K Rb Cs Fr – Group 1
(Happy Little Naughty Kids Rub Cake Freely)
👉 Be Mg Ca Sr Ba Ra – Group 2
(Be My Cute Sister’s Best Relative)
👉 F Cl Br I At Ts – Halogens
(Father Calls Brother Inside At Tea Shop)
Students enjoy studying when they understand through shortcuts.
⭐ Why Should Students Use the Periodic Table Daily?
Because it makes chemistry simple.
When you know the table:
✔ You can predict reactions
✔ You score better in exams
✔ You don’t need to memorize everything
It is like having a cheat-sheet of chemistry!
⭐ Conclusion
The periodic table is not just a display chart—it is a scientific tool that helps students from class 9th to 12th understand chemistry better. When students begin learning elements using the periodic table, everything becomes easier—formulas, reactions, bonding, trends, valency, and more.
So whether you want to excel in school exams, NEET, JEE or olympiads, the periodic table is your strongest foundation.